

Redefining drug delivery.
NaDeNo’s drug delivery platform enables targeted delivery of hard-to-deliver drugs.
Our drug delivery platform enable novel and differentiated therapeutics, and we are open to partnerships and co-developments with companies in need of a versatile drug delivery technology for hard-to-deliver drugs.
Numerous applicable drug classes
Successful encapsulation of numerous drug classes across indications with high unmet need and commercial potential.
Sustainable & scalable manufacturing
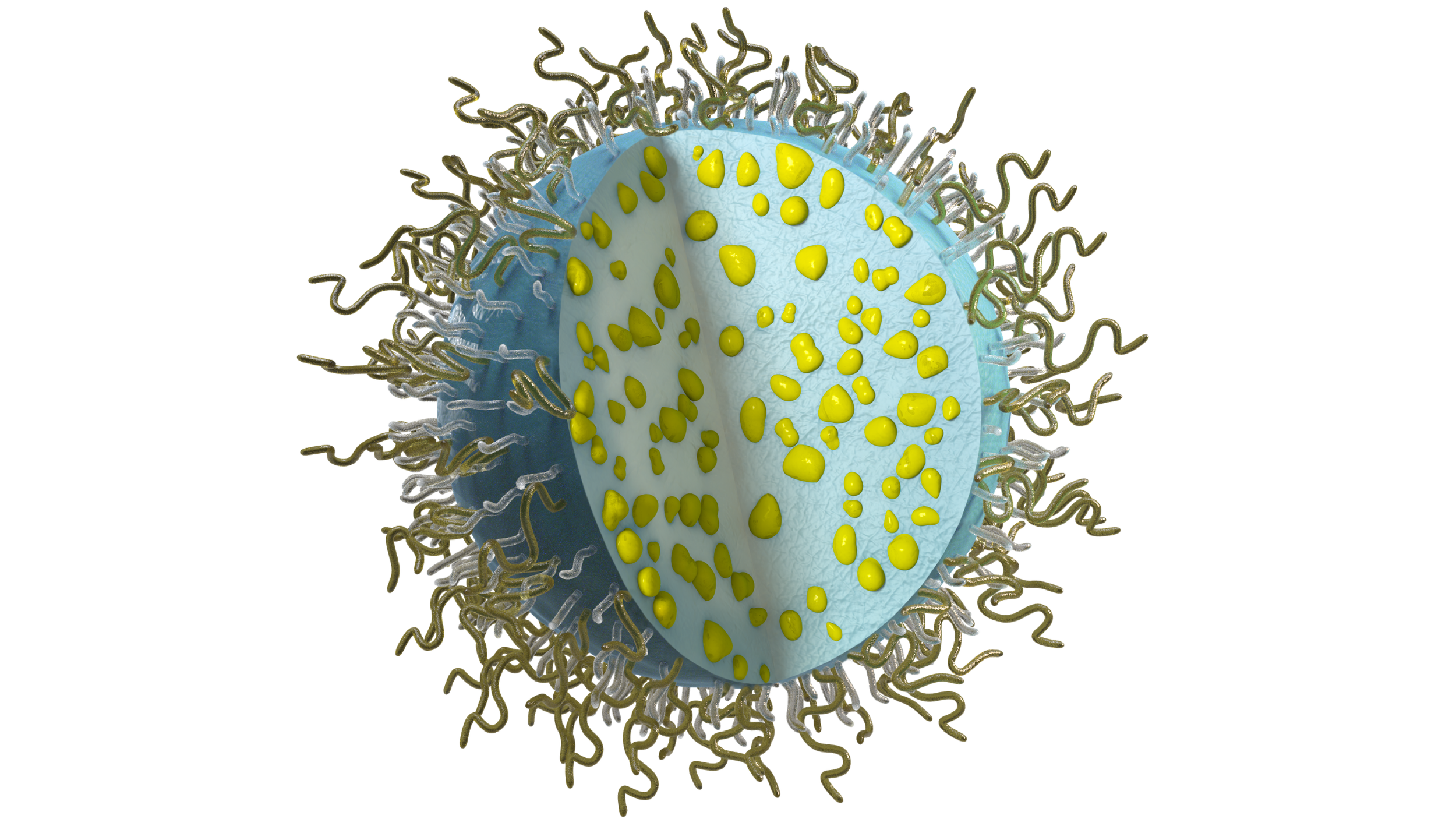
NaDeNo’s nanoparticles encapsulate the drug in a patented 1-step process without any chemical modification of the drug.
Adaptive surface targeting
By functionalizing the surface, the nanoparticles can achieve various forms of chemical and/or biological targeting.
Retention and controlled release
Proven retention following various local administration routes, combined with controlled release for sustained efficacy.
Lead asset
PACAB-002 to prevent recurrence in ovarian cancer

NaDeNo’s platform-enabled lead candidate PACAB-002 will start clinical investigation in a Phase I trial beginning early 2027. PACAB-002 is intended to eradicate peritoneal micrometastases for patients with high-grade serous ovarian cancer (HGSOC), the leading cause of high recurrence and poor survival.
